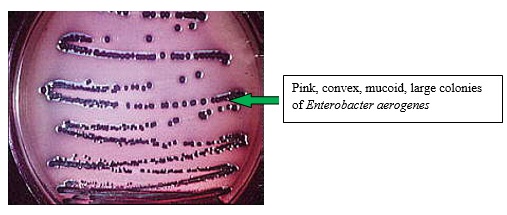
Theory:
Water is unsafe to consume if gas producing lactose fermenting microorganisms present in water. A gas formation may be due to non-coliform bacteria such as Clostridium perfringens, which are gram-positive, and also it may due to gram-negative coliform. The presence of Coliform bacteria is an indication of the unsuitability of water. E.colithat is one of coliform are usually resided in human intestine and known to be fecal in origin. To confirm the presence of gram-negative lactose fermenters in water, the confirmed test is carried out.
Streak Plate Method
First sufficient amount of agar was poured into a sterile petri dish. Next, this was allowed to solidify. Then a sterilized inoculating needle was dipped in the Durham tube containing test tube. Next, the agar plate was streaked to separate the two types of coliforms. Finally, this was incubated at 370C.
Labels:
Microbiology Practicals
Spread Plate Method
Sufficient amount of agar was poured into a sterile petri dish. Next, this was allowed to solidify. 0.1ml of the fermented water samples were transferred on to the agar surface. A spreader was dipped in 70% alcohol and was flamed. Finally, the sample was spread over the surface of the agar then lids were closed.
Labels:
Microbiology Practicals
Pour Plate Method
First 1ml of the fermented water samples were transferred into sterile Petri dishes, using a sterile pipette. 15ml of Eosin methylene blue agar was poured into the Petri dish and rotate the Petri dish while on the table to mix the water sample with the agar. Next, this was allowed to solidify. Finally, this incubated at 370C.
Labels:
Microbiology Practicals
Obtaining Pure Microbial Culture
Streak plate or spread plate
This will be discussed under the section “practicals”.
Dilution plating
A series of test tubes containing 9 ml of sterile distilled water or 0.85% NaCl, peptone water and Ringer's solution has to be prepared which are called dilution blanks.1ml of the initial or original sample is transferred into the 1st test tube (10-1).Shake.Next 1ml of 10-1 solution is transferred into the next test tube (10-2). This procedure is carried out for another 5/6 test tubes. A sterile pipette is used to transfer solutions at each step. Then a pour plate is made for each and every dilution. (will be discussed at practicals section).
 |
Dilution plating
|
Labels:
Microbiology Basics
Culturing Microorganisms
There are various culture media. Mixed culture, pure culture and in vitro ( cultured in containers), in vivo (grown in live animal and plant tissue).Culture medium basically requires a carbon source, nitrogen source, macronutrients, micronutrients, growth factors, gelling agents, antibiotics and dyes. There are two gelling agents agar and gelatin.
Agar is made out of red algae which have a melting point of 85 Celsius and a complex mixture of polysaccharides extracted from red algae. It is insoluble in cold water and sets between 32-39 Celsius. Gelatin is made out of hooves of animals. Thus high in protein. Melting point 30-35 celsius and readily digests by some bacteria.
Antibiotics are generally added before pouring into plates to inhibit a specific group of microorganisms. Since streptomycin is heat resistant it is added before heating whereas penicillin is heat sensitive thus added after heating.
Natural media
First used by Leeuwenhoek. This medium is prepared using peppercorns in water, hay in water and also meat in water. These are called infusions. Since the composition is not known it is difficult to reproduce.Eg; PDA, NA, Malt extract.
Artificial media
First used by pasture. Can be reproduced.Eg; Capex-dox nutrient.
- NaNO3 2g
- K2HPO4 1g
- MgSO4.7H2O 0.5g
- KCl 0.5g
- FeSO4 0.01g
- Sucrose 30g
- Agar 20g
- Distilled water 1L
Basal media
Both natural and synthetic media.This suite for broad ranges of nutritionally undemanding groups of microorganisms.Eg; Cook’s no 2.
Selective media
A media that favor the growth of one type of specific microorganism while inhibiting the growth of undesirable microorganisms. By adding or removing of certain nutritional substances can separate one organism from another.
Eg; Coliform bacteria, Salmonella typhi.
Differential media
A culture media that distinguish different types of microorganisms by their metabolism of media components and colony appearance.Eg; Eosin Methylene blue agar, Yeast mannitol agar.
Suppressive media
Suppression of bacteria and encouragement of fungal growth.
Eg; Martin’s medium. This medium contains the antibiotic streptomycin and the dye rose Bengal. Rose Bengal suppresses bacterial growth as a result fungi grow.
Labels:
Microbiology Basics
Gram Staining Procedure
Gram stain is a method of differential staining. Gram stain was discovered and introduced by Christian Gram in 1884. This divides bacteria into two groups, gram positive and gram negative, depending on the presence of peptidoglycan in their cell wall. Bacteria lacking peptidoglycan termed as gram negatives and gram positives are the bacteria who contain peptidoglycan. Peptidoglycan can absorb dyes hence the presence of peptidoglycan gives color.
Gram staining method has two particular dyes. Primary dye is the ammonium oxalate crystal violet which stains all cells with purple. Mordant is a substance that strengthens the union between the dye and the substrate or fixes coloring material. Gram’s iodine solution used as the mordant. Decolorizing agent 95% alcohol removes the primary stain from gram-negative cells and gram positives retain the primary dye. Safranin a secondary dye is used to stain the colorless gram-negative cells. Hence gram-positive cells appear in purple whereas gram-negative cells show a color of pink to red. This differential behavior has been explained by; the cell wall composition of gram negatives and positives and differential permeability to alcohol insoluble dye, iodine complex.
Gram staining method has two particular dyes. Primary dye is the ammonium oxalate crystal violet which stains all cells with purple. Mordant is a substance that strengthens the union between the dye and the substrate or fixes coloring material. Gram’s iodine solution used as the mordant. Decolorizing agent 95% alcohol removes the primary stain from gram-negative cells and gram positives retain the primary dye. Safranin a secondary dye is used to stain the colorless gram-negative cells. Hence gram-positive cells appear in purple whereas gram-negative cells show a color of pink to red. This differential behavior has been explained by; the cell wall composition of gram negatives and positives and differential permeability to alcohol insoluble dye, iodine complex.
This method is to distinguish gram negatives and gram-positive bacteria. This comparison depends on the presence of peptidoglycan in the cell wall.
Labels:
Microbiology Basics
Subscribe to:
Comments (Atom)
Popular Posts
-
Gram stain is a method of differential staining. Gram stain was discovered and introduced by Christian Gram in 1884. This divides bacteria ...
-
Nuts are regularly prone to chemical changes. Fats and oils are the major constituents of nuts. Rancidity causes due to high content of oil ...
-
There are various culture media. Mixed culture, pure culture and in vitro ( cultured in containers), in vivo (grown in live animal and plan...
-
Theory The routine microbial examination of water to determine its portability is not based on the isolation and identification of path...
-
Theory: Water is unsafe to consume if gas producing lactose fermenting microorganisms present in water. A gas formation may be due to non-...
-
The main causation for the spoilage of food is by microbes. There are three types of microorganisms that cause food spoilage. Those are bact...
-
Observation of living cells follows below procedures. Negative Staining Microorganism will not stain in this method, but the medium...
-
Physical damages on food items is caused through wounds, Stress conditions such as water stress, nutrimental stresses etc., extreme conditio...
-
Streak or spread plate Dilution plating Dilution culturing Streak plate or spread plate This will be discussed under the section “...
-
Microorganisms are invisible to the naked eye. Therefore for the purpose of studying, they are cultured. Staining will kill microorganism...
Microbiology Basics
Microbiology Tests
Food Microbiology
- Effect of Interactions on Food Microbes
- Food Spoilage
- Microbial Activity on Food Spoilage
- Chemical Changes on Food Spoilage
- Effect of Physical Damage on Food Spoilage
- Freezer Burn in Food Spoilage
- Staling Effect for Food Spoilage
- Contaminating Micro Flora Composition Factors
- Factors Determining the Composition of the Contaminating Micro Flora
- Effect of Nutrient Compositionon Food Microbes
- Spoilage of Canned Foods in Microbiology Aspect